Ask Science
Ask a science question, get a science answer.
Community Rules
Rule 1: Be respectful and inclusive.
Treat others with respect, and maintain a positive atmosphere.
Rule 2: No harassment, hate speech, bigotry, or trolling.
Avoid any form of harassment, hate speech, bigotry, or offensive behavior.
Rule 3: Engage in constructive discussions.
Contribute to meaningful and constructive discussions that enhance scientific understanding.
Rule 4: No AI-generated answers.
Strictly prohibit the use of AI-generated answers. Providing answers generated by AI systems is not allowed and may result in a ban.
Rule 5: Follow guidelines and moderators' instructions.
Adhere to community guidelines and comply with instructions given by moderators.
Rule 6: Use appropriate language and tone.
Communicate using suitable language and maintain a professional and respectful tone.
Rule 7: Report violations.
Report any violations of the community rules to the moderators for appropriate action.
Rule 8: Foster a continuous learning environment.
Encourage a continuous learning environment where members can share knowledge and engage in scientific discussions.
Rule 9: Source required for answers.
Provide credible sources for answers. Failure to include a source may result in the removal of the answer to ensure information reliability.
By adhering to these rules, we create a welcoming and informative environment where science-related questions receive accurate and credible answers. Thank you for your cooperation in making the Ask Science community a valuable resource for scientific knowledge.
We retain the discretion to modify the rules as we deem necessary.
view the rest of the comments
no. this is what Heisenberg's uncertainty principle is about. we cannot know both a quantum particle's location and it's velocity with high accuracy at the same time. if we know with high accuracy how fast it's moving, we can only have a very vague guess of where it is, or vice versa
Usually for typical conductors I think we can learn enough about the position and momentum of our electrons to keep track of them.
Not overly precisely but enough to not confuse them with each other.
A good way to see this is that we can simulate electrons moving through states in a conductor with good accuracy, without a need to go into full quantum mechanical descriptions, in an almost classical simulation.
Of course the position and speed we are tracking there is not a typical blurred point, it will be a complicated wave spanning many neighbouring atoms in size, with different electrons being at different positions around those atoms.
But you can know which electrons are in what loose region with what distribution, and follow them through interactions where they move to different regions or change the shape of their wave. Depending on your conductor the spread may even go down to single-atom-scales in some extremes.
Measuring all electrons in a real conductor enough to tell after some time which end of it any one electron ended up at, would probably change its properties slightly due to the measurements, but done correctly it should definitely still be behaving like a normal conductor.
Here is an example of a particularly low-speed localized electron in a typical material:
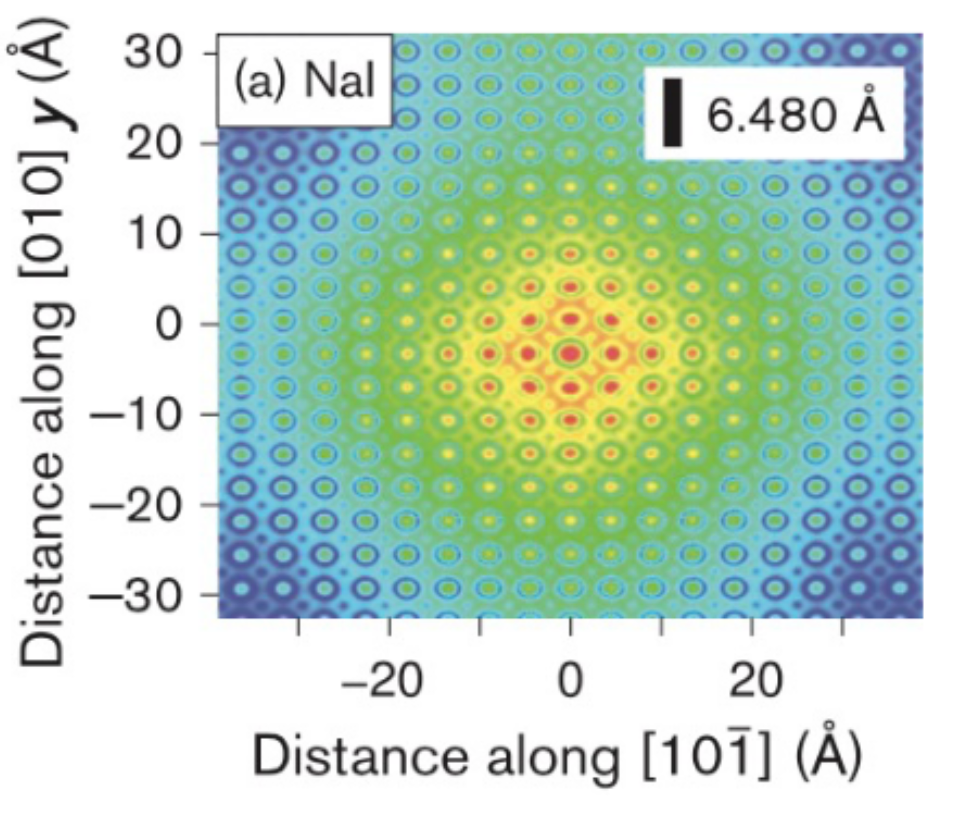
Every electron will have a patterned distribution like that. If you naively tried to measure electrons at some spot, there would be thousands strongly overlapping there, you would mess everything up. But there is no issue checking if this wide shape as shown has an electron occupying it or not.